Understanding the Difference Between Allylic and Vinylic Carbons is essential in organic chemistry because these carbon positions strongly influence reactivity, stability, and reaction mechanisms. An allylic carbon is the carbon atom adjacent to a double bond (not part of the double bond), whereas a vinylic carbon is a carbon atom that directly participates in a carbon–carbon double bond.
Both play crucial roles in radical reactions, substitution, addition reactions, and resonance stabilization, making them important for students and professionals working with alkenes, polymer chemistry, and reaction mechanisms.
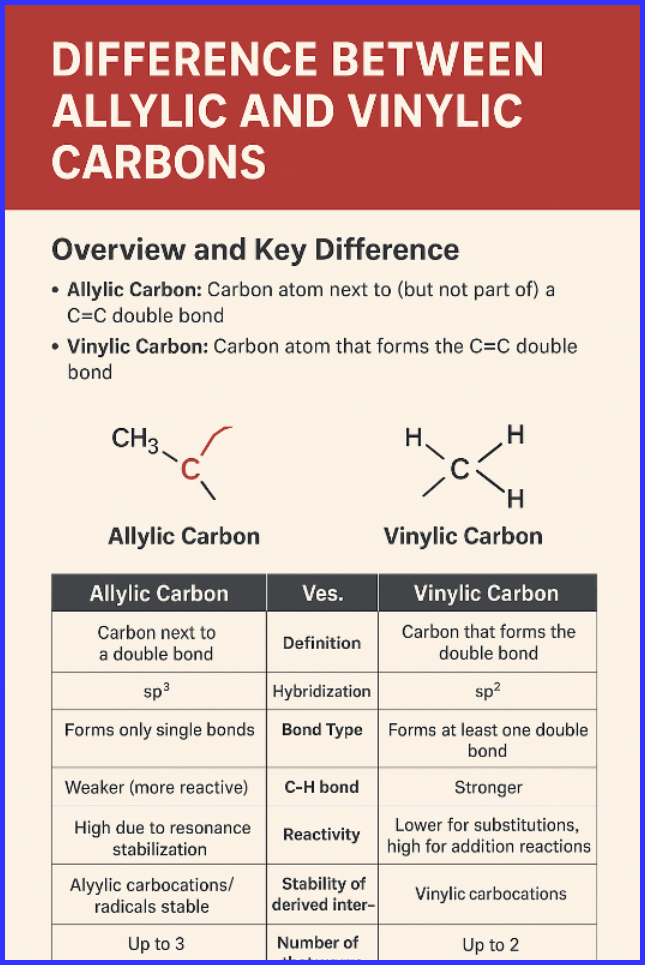
Overview and Key Difference
- Allylic Carbon: Carbon atom next to (but not part of) a C=C double bond.
- Vinylic Carbon: Carbon atom that forms the C=C double bond itself.
- Key Difference:
Allylic carbons are sp³-hybridized and indirectly connected to a double bond, while vinylic carbons are sp²-hybridized and directly part of the double bond.
What is an Allylic Carbon?
An allylic carbon is the carbon atom adjacent to a double bond. It is not part of the double bond itself but is directly bonded to one of the double-bonded carbons.
Structure Example
- General formula: CH₃–CH=CH₂
Here, the CH₃ carbon is the allylic carbon.
Hybridization
- sp³ hybridized
- Single-bonded to an sp² carbon.
Characteristics
- Has allylic hydrogens, which are more reactive because the C–H bond is weaker.
- Shows resonance stabilization when forming allylic carbocations, radicals, or anions.
- Commonly participates in:
- Allylic substitution reactions
- Radical halogenation
- SN1/SN2 reactions (better than normal alkyl carbons due to resonance)
Why Allylic Carbons Are More Reactive
The adjacent double bond can delocalize electrons, making:
- Allylic carbocations highly stabilized.
- Allylic radicals more stable than typical alkyl radicals.
This enhanced stability leads to high reactivity at the allylic position.
What is a Vinylic Carbon?
A vinylic carbon is a carbon directly involved in a carbon–carbon double bond (C=C).
Structure Example
- General alkene formula: CH₂=CH₂
Here, both CH₂ carbons are vinylic carbons.
Hybridization
- sp² hybridized
- Participates in one sigma bond + one pi bond.
Characteristics
- High electron density because of the double bond.
- Does not undergo SN1 or SN2 reactions (vinylic carbocations are unstable).
- Participates mainly in electrophilic addition reactions.
- Can be part of linear, branched, or cyclic alkenes.
Special Case
In cumulenes like CH₂=C=CH₂, all three central carbon atoms are vinylic due to multiple connecting double bonds.
Summary Table: Key Difference between Allylic and Vinylic Carbons
| Feature | Allylic Carbon | Vinylic Carbon |
| Definition | Carbon next to a double bond | Carbon that forms the double bond |
| Position Relative to C=C | Indirectly connected | Directly connected |
| Hybridization | sp³ | sp² |
| Bond Type | Forms only single bonds | Forms at least one double bond |
| C–H Bond Strength | Weaker (more reactive) | Stronger |
| Reactivity | High due to resonance stabilization | Lower for substitutions; high for addition reactions |
| Stability of Intermediates | Allylic carbocations/radicals are stable | Vinylic carbocations are very unstable |
| Number of Hydrogens Possible | Up to 3 | Up to 2 |
| Typical Reactions | Allylic substitution, radical halogenation | Electrophilic addition to C=C |
| Electron Density | Lower than vinylic | Higher at the double bond |
Summary
The Difference Between Allylic and Vinylic Carbons lies in how each relates to a double bond. An allylic carbon is next to the C=C bond and is sp³-hybridized, showing higher reactivity due to resonance stabilization. A vinylic carbon, on the other hand, is part of the double bond and is sp²-hybridized, making it central to electrophilic addition reactions.
Understanding this distinction helps predict reactivity trends, stability of intermediates, and mechanisms in organic chemistry. This makes the Difference Between Allylic and Vinylic Carbons extremely important for students, chemists, and anyone working with alkenes and functional groups.
Read Next: