The key difference between covalent and noncovalent bonds is that covalent bonds involve the sharing of electrons between atoms, whereas noncovalent bonds form without sharing electrons—either by complete electron transfer or by weak intermolecular forces. This fundamental difference determines how molecules form, interact, and behave in chemical and biological systems.
Both types of bonds play essential roles in chemistry, biological structures, drug interactions, and material science.
Before diving deeper, it’s important to understand how these bonds exist in nature:
- Covalent bonds hold atoms together within molecules (intramolecular).
- Noncovalent bonds create attractions between molecules or between different parts of the same large molecule (intermolecular).
What Are Covalent Bonds?
A covalent bond is a strong chemical bond formed when two atoms share one or more pairs of electrons. This type of bonding helps atoms achieve a stable electronic configuration, usually mimicking a full outer shell.
Characteristics of Covalent Bonds
- Usually form between nonmetal atoms with similar electronegativity.
- Create stable molecules such as water (H₂O) and methane (CH₄).
- Strength is higher compared to noncovalent bonds.
- Defined by bond length, bond energy, and bond angle.
Types of Covalent Bonds
1. Polar Covalent Bonds
- Form when the electronegativity difference is 0.4–1.7.
- Electrons are shared unequally.
- Example: H–Cl, H₂O
Oxygen pulls electrons more strongly → molecule becomes polar.
2. Nonpolar Covalent Bonds
- Form when electronegativity difference is < 0.4.
- Electrons shared equally.
- Example: O₂, N₂, CH₄
3. Single, Double, and Triple Bonds
- Single bond → 1 electron pair shared (e.g., H–H)
- Double bond → 2 pairs shared (e.g., O=O)
- Triple bond → 3 pairs shared (e.g., N≡N)
Structure of Covalent Bonds
The structure of molecules with covalent bonds depends on:
- Electron pair repulsion (VSEPR theory)
- Hybridization (sp, sp², sp³)
Example structures:
- H₂O: bent shape due to lone pairs
- CO₂: linear
- CH₄: tetrahedral
What Are Noncovalent Bonds?
Noncovalent bonds are weak chemical interactions that occur without electron sharing. They are essential in biological systems, protein folding, DNA stability, and molecular recognition.
Types of Noncovalent Bonds
1. Ionic Bonds
- Form by complete electron transfer.
- Resulting ions attract each other via electrostatic forces.
- Example: Na⁺ + Cl⁻ → NaCl
2. Hydrogen Bonds
- Weak attraction between a hydrogen atom and an electronegative atom (N, O, F).
- Stronger than van der Waals forces but weaker than covalent bonds.
- Examples:
- Water molecules bonding with each other
- DNA base pairing (A–T and G–C)
3. Van der Waals Interactions
- Weak, temporary attractions due to fluctuating electron clouds.
- Occur in nonpolar molecules.
- Examples:
- Attraction between noble gas atoms
- Gecko feet adhesion
Structure of Noncovalent Bonds
Noncovalent structures are temporary and dynamic. They stabilize:
- Protein folding
- DNA double helix
- Enzyme–substrate interactions
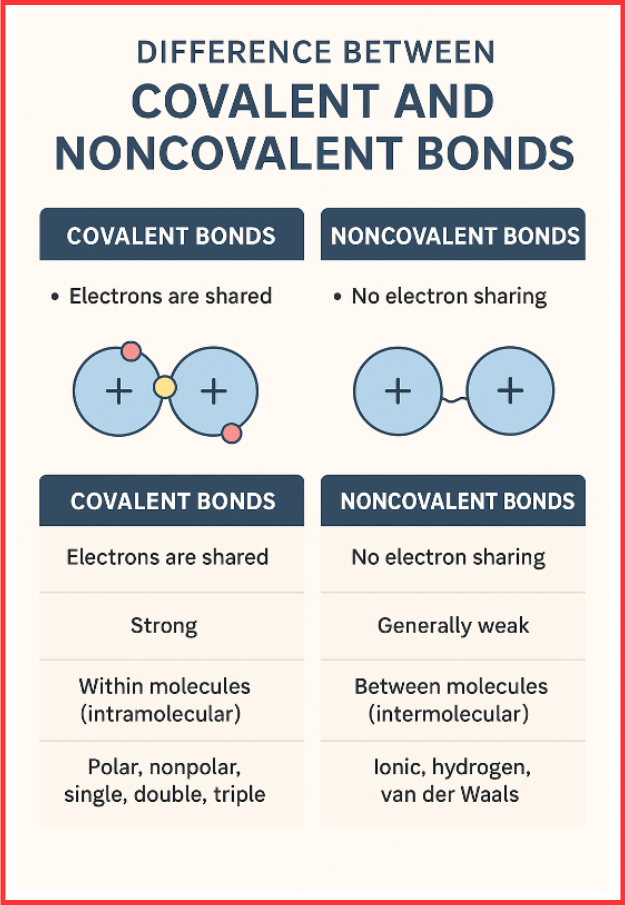
Difference Between Covalent and Noncovalent Bonds (Tabular Comparison)
| Feature | Covalent Bonds | Noncovalent Bonds |
| Electron Interaction | Electrons are shared | No electron sharing; may involve electron transfer |
| Bond Strength | Strong | Generally weak |
| Occurrence | Within molecules (intramolecular) | Between molecules (intermolecular) |
| Types | Polar, nonpolar, single, double, triple | Ionic, hydrogen, van der Waals |
| Example | H₂O, CH₄, O₂ | NaCl (ionic), H-bonds in water, van der Waals in gases |
| Electronegativity Role | Determines polarity | Determines ion formation or attraction |
| Function | Creates stable molecules | Stabilizes molecular interactions |
Summary – Covalent vs Noncovalent Bonds
Covalent and noncovalent bonds are essential for forming and stabilizing matter. The difference between covalent and noncovalent bonds lies in their electron interactions: covalent bonds involve electron sharing, while noncovalent bonds rely on electrostatic attractions or intermolecular forces. Understanding both types is crucial for studying chemistry, biology, materials, and molecular sciences.
In conclusion, the difference between covalent and noncovalent bonds helps explain how molecules form, interact, and function in both chemical and biological environments.
Reference:
1. “Van Der Waals Forces.” Chemistry LibreTexts, Libretexts, 15 Aug. 2020,
Read Next: