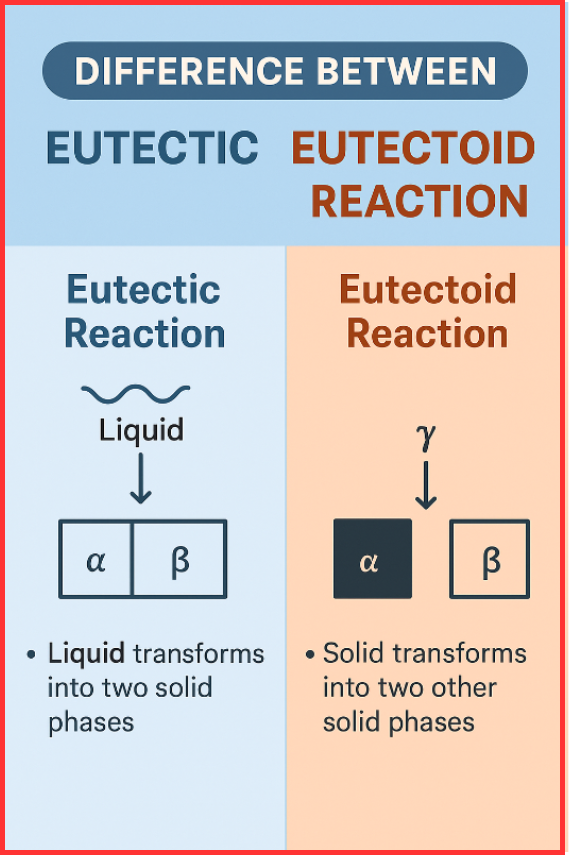
The difference between eutectic and eutectoid reaction is that a eutectic reaction transforms a liquid into two solid phases simultaneously, whereas a eutectoid reaction transforms one solid phase into two different solid phases at the same time. Both are three-phase transformations driven by temperature changes in alloy systems.
Phase transformations play a vital role in metallurgy and material science. Among these, eutectic and eutectoid reactions are especially important because they define how alloys behave during heating and cooling.
What is a Eutectic Reaction?
A eutectic reaction occurs when a liquid phase transforms into two solid phases simultaneously upon cooling.
Key Features of Eutectic Reaction
- A liquid phase → two solid phases (L → α + β)
- Occurs at a fixed temperature called the eutectic temperature
- A eutectic mixture has the lowest melting point among all compositions of that system
- It is a three-phase equilibrium (liquid + two solid phases)
Understanding Eutectic Systems
A eutectic system is a homogeneous mixture that:
- Melts at a lower temperature than any individual component
- Solidifies into two solid phases at the same temperature
Example:
In the lead–tin (Pb–Sn) binary alloy system, the liquid alloy transforms into α-solid (Pb-rich) and β-solid (Sn-rich) at the eutectic point.
Why Eutectic Reactions Matter
- They help in designing low-melting alloys (e.g., solder)
- They produce fine, layered microstructures with desirable mechanical properties
What is a Eutectoid Reaction?
A eutectoid reaction is a solid-state transformation where one solid phase decomposes into two new solid phases simultaneously.
Key Features of Eutectoid Reaction
- A solid phase → two solid phases (γ → α + β)
- Occurs at a fixed eutectoid temperature
- It is also a three-phase equilibrium, but with all phases solid
Most Common Example: Iron–Carbon System
The best-known eutectoid reaction occurs in steel:
Austenite (γ) → Ferrite (α) + Cementite (Fe₃C)
This transformation forms pearlite, a layered microstructure responsible for the strength of many steel grades.
Importance of Eutectoid Reactions
- Controls heat treatments such as annealing, normalizing, and hardening
- Determines mechanical properties like hardness, ductility, and strength
Difference Between Eutectic and Eutectoid Reaction (Table)
| Aspect | Eutectic Reaction | Eutectoid Reaction |
| Initial Phase | Liquid | Solid |
| Final Phases | Two solids | Two solids |
| Transformation Type | L → α + β | γ → α + β |
| Temperature | Eutectic temperature | Eutectoid temperature |
| Typical Example | Lead–tin alloy | Pearlite formation in steel |
| Application | Soldering alloys, casting | Heat treatment, steel strengthening |
| Nature of Change | Liquid-to-solid transformation | Solid-to-solid transformation |
Examples for Better Understanding
Eutectic Example
- Pb–Sn solder: Liquid alloy → α (Pb-rich) + β (Sn-rich)
Eutectoid Example
- Steel: Austenite → ferrite + cementite (pearlite)
Summary – Eutectic vs Eutectoid Reaction
The main difference between eutectic and eutectoid reaction is the phase that undergoes transformation.
- A eutectic reaction converts a liquid into two solid phases.
- A eutectoid reaction converts a solid into two new solid phases.
Both reactions are crucial in alloy design, manufacturing, and controlling material properties. Understanding the difference between eutectic and eutectoid reaction helps engineers optimize hardening, welding, soldering, and casting processes.
Reference:
1. Soni, Prem Kumar. “Eutectic, Eutectoid, Peritectoid, Peritectic.” SlideShare, 5 Aug. 2018,
Read Next:
- Difference Between Px Py and Pz Orbitals
- Difference Between Covalent and Noncovalent Bonds
- Difference Between Aldohexose and Ketohexose
- Difference Between Percent Yield and Percent Recovery
- Difference Between Allylic and Vinylic Carbons
- Difference Between Acetic Acid and Acetate
- Difference Between Normal Shampoo and Conditioning Shampoo