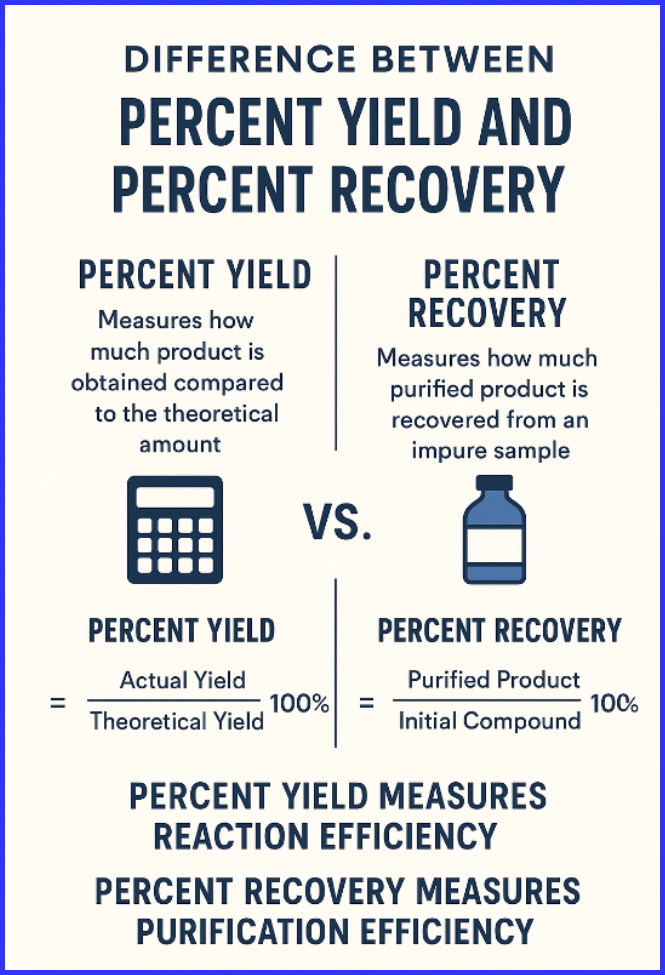
The difference between percent yield and percent recovery is that percent yield measures how much product is obtained compared to the theoretical amount, while percent recovery measures how much purified product is recovered from an impure sample. Percent yield evaluates reaction efficiency, whereas percent recovery evaluates purification efficiency.
Understanding the difference between percent yield and percent recovery is essential in chemistry, especially in synthesis and purification experiments. Both are percentage-based calculations, but they describe two different aspects of laboratory efficiency. Percent yield focuses on the actual product formed, while percent recovery focuses on the purified product obtained after removing impurities.
What Is Percent Yield?
Percent yield expresses how efficiently a chemical reaction produces the desired product. It is the ratio of the actual yield (product obtained in the lab) to the theoretical yield (product expected from stoichiometric calculations).
In most experiments, the actual yield is lower due to:
- Incomplete reactions
- Loss during filtration or transfer
- Side reactions
- Impurities
A percent yield of 100% means perfect efficiency, although this is extremely rare.
How to Calculate Percent Yield
Percent Yield Formula
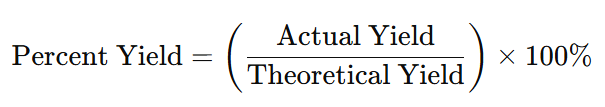
Note: Sometimes percent yield may exceed 100% due to impurities or measurement errors.
Percent Yield Example
When 25 g of CaCO₃ is heated for decomposition, 12.3 g of CaO is obtained.
Reaction:
CaCO3→CaO+CO2
- Molar mass of CaCO₃ = 100 g/mol
- Molar mass of CaO = 56 g/mol
Step 1: Theoretical Yield
From 100 g CaCO₃ → 56 g CaO
From 25 g CaCO₃:
Theoretical Yield=(56/100)×25=14 g
Step 2: Percent Yield
Percent Yield=(12.3/14)×100=87.86%
So, the percent yield = 87.86%.
What Is Percent Recovery?
Percent recovery expresses how much purified product is obtained after purification processes like recrystallization. It is used mainly in organic chemistry to determine how effectively impurities were removed.
Purification steps like recrystallization often cause some loss, so recovery never reaches 100%.
How to Calculate Percent Recovery
Percent Recovery Formula

This calculation helps evaluate:
- Purification efficiency
- Product loss during washing or filtering
- Effectiveness of solvent selection
Percent Recovery Example
If 20 g of an impure organic solid is taken for recrystallization and 15.5 g of purified solid is recovered at the end:
Percent Recovery=(15.5/20)×100=77.5%
So, the percent recovery = 77.5%.
Similarities Between Percent Yield and Percent Recovery
Both calculations:
- Are percentages
- Use a ratio multiplied by 100
- Measure laboratory efficiency
- Are influenced by experimental accuracy and technique
Key Difference Between Percent Yield and Percent Recovery (Tabular Form)
| Feature | Percent Yield | Percent Recovery |
| Definition | Ratio of actual yield to theoretical yield | Ratio of purified product to original impure product |
| Purpose | Measures efficiency of a chemical reaction | Measures efficiency of purification |
| Used In | Synthesis / reaction calculations | Recrystallization and purification |
| Formula | (Actual Yield ÷ Theoretical Yield) × 100 | (Purified Product ÷ Initial Impure Product) × 100 |
| Value >100%? | Possible due to impurities | Rare but possible due to solvent retention |
Conclusion
The difference between percent yield and percent recovery is mainly based on what each value measures. Percent yield evaluates how efficiently a reaction produces the desired product, while percent recovery evaluates how effectively a product is purified. Understanding both concepts helps students and chemists improve reaction design, optimize purification steps, and enhance overall laboratory efficiency.
Read Next: