The key difference between aldohexose and ketohexose is that aldohexoses contain a formyl (–CHO) group at carbon 1, whereas ketohexoses contain a ketone (>C=O) group usually at carbon 2. Both are six-carbon monosaccharides, but their functional groups and stereochemical arrangements make them structurally and functionally different.
Understanding the difference between aldohexose and ketohexose is essential in carbohydrate chemistry, biochemistry, food science, and metabolic biology.
What is an Aldohexose?
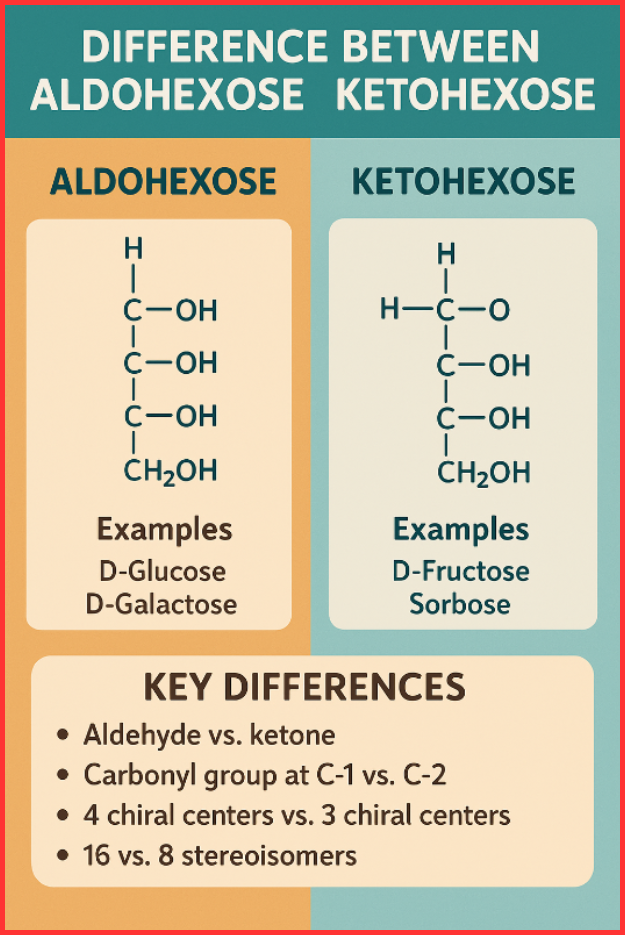
An aldohexose is a six-carbon monosaccharide that contains an aldehyde (–CHO) group at carbon 1. Aldohexoses are special types of aldoses, and their linear structure can be written as:
Structure of Aldohexose
H–C(=O)–(CHOH)₄–CH₂OH
- Carbonyl carbon is at position 1
- Contains four chiral centers
- Gives rise to 16 stereoisomers (8 enantiomeric pairs)
Examples of Aldohexose
- D-Glucose (most abundant natural sugar)
- D-Galactose
- D-Mannose
- Allose, Altrose, Talose, Idose (less common)
Why Aldohexoses are Important
Aldohexoses play major roles in:
- Energy metabolism (glucose)
- Glycoprotein synthesis
- Cellular respiration pathways
What is a Ketohexose?
A ketohexose is a six-carbon monosaccharide that contains a ketone (>C=O) group, usually at carbon 2. Ketohexoses are a subclass of ketoses.
Structure of Ketohexose
CH₂OH–C(=O)–(CHOH)₃–CH₂OH
- Carbonyl carbon is at position 2 (rarely at 3)
- Contains three chiral centers
- Has 8 stereoisomers (4 enantiomeric pairs)
Examples of Ketohexose
- D-Fructose (fruit sugar; most common)
- Sorbose
- Tagatose
- Psicose
Why Ketohexoses Matter
Ketohexoses are essential in:
- Energy metabolism (fructose)
- Food sweeteners
- Metabolic pathway intermediates
Aldohexose vs Ketohexose: Key Differences
Below is a clear comparison of the difference between aldohexose and ketohexose for easy understanding.
1. Functional Group
- Aldohexose: Contains an aldehyde (–CHO)
- Ketohexose: Contains a ketone (>C=O)
2. Position of Carbonyl Group
- Aldohexose: Carbonyl at C-1
- Ketohexose: Carbonyl at C-2 (sometimes C-3)
3. Number of Chiral Centers
- Aldohexose: 4 chiral centers
- Ketohexose: 3 chiral centers
4. Number of Stereoisomers
- Aldohexose: 16
- Ketohexose: 8
5. Common Biological Example
- Aldohexose: D-Glucose
- Ketohexose: D-Fructose
Difference Between Aldohexose and Ketohexose (Table)
| Feature | Aldohexose | Ketohexose |
| Functional Group | Aldehyde (–CHO) | Ketone (>C=O) |
| Carbonyl Position | Carbon 1 | Carbon 2 (or 3 rarely) |
| Number of Carbons | 6 | 6 |
| Chiral Centers | 4 | 3 |
| Stereoisomers | 16 | 8 |
| Common Example | Glucose | Fructose |
| Oxidation | Easily oxidized | Less reactive toward oxidation |
| Cyclic Structure | Forms pyranose rings mostly | Forms furanose rings mostly |
Examples With Structures
Example of Aldohexose: D-Glucose
- Carbonyl group at C-1
- Forms pyranose ring
- Present in blood sugar and energy metabolism
Example of Ketohexose: D-Fructose
- Carbonyl group at C-2
- Generally forms furanose ring
- Found in fruits, honey, and sucrose
Summary – Aldohexose vs Ketohexose
Aldohexoses and ketohexoses are both hexose monosaccharides, but the difference between aldohexose and ketohexose lies in their functional groups. Aldohexoses have an aldehyde group at carbon 1, while ketohexoses have a ketone group usually at carbon 2.
Aldohexose examples include glucose, whereas ketohexose examples include fructose.
In conclusion, understanding the difference between aldohexose and ketohexose helps clarify their role in biological reactions, metabolic pathways, and structural variations in carbohydrates.
Reference:
1. “14.3: Important Hexoses.” Chemistry LibreTexts, Libretexts, 23 Feb. 2019
2. “Hexose.” Wikipedia, Wikimedia Foundation, 2 June 2020,
Read Next: